Costacurta produce tele e reti metalliche per celle a diaframma utilizzate nella
produzione di soda caustica (processo cloro-soda).
Il processo cloro-soda è un processo elettrochimico che permette, partendo da una soluzione di cloruro di sodio (salamoia), di ottenere per elettrolisi cloro gassoso, soda caustica e idrogeno. La produzione di cloro e soda può avvenire mediante celle a diaframma, celle a membrana o celle a mercurio.
Hai una richiesta specifica?
Contattaci ora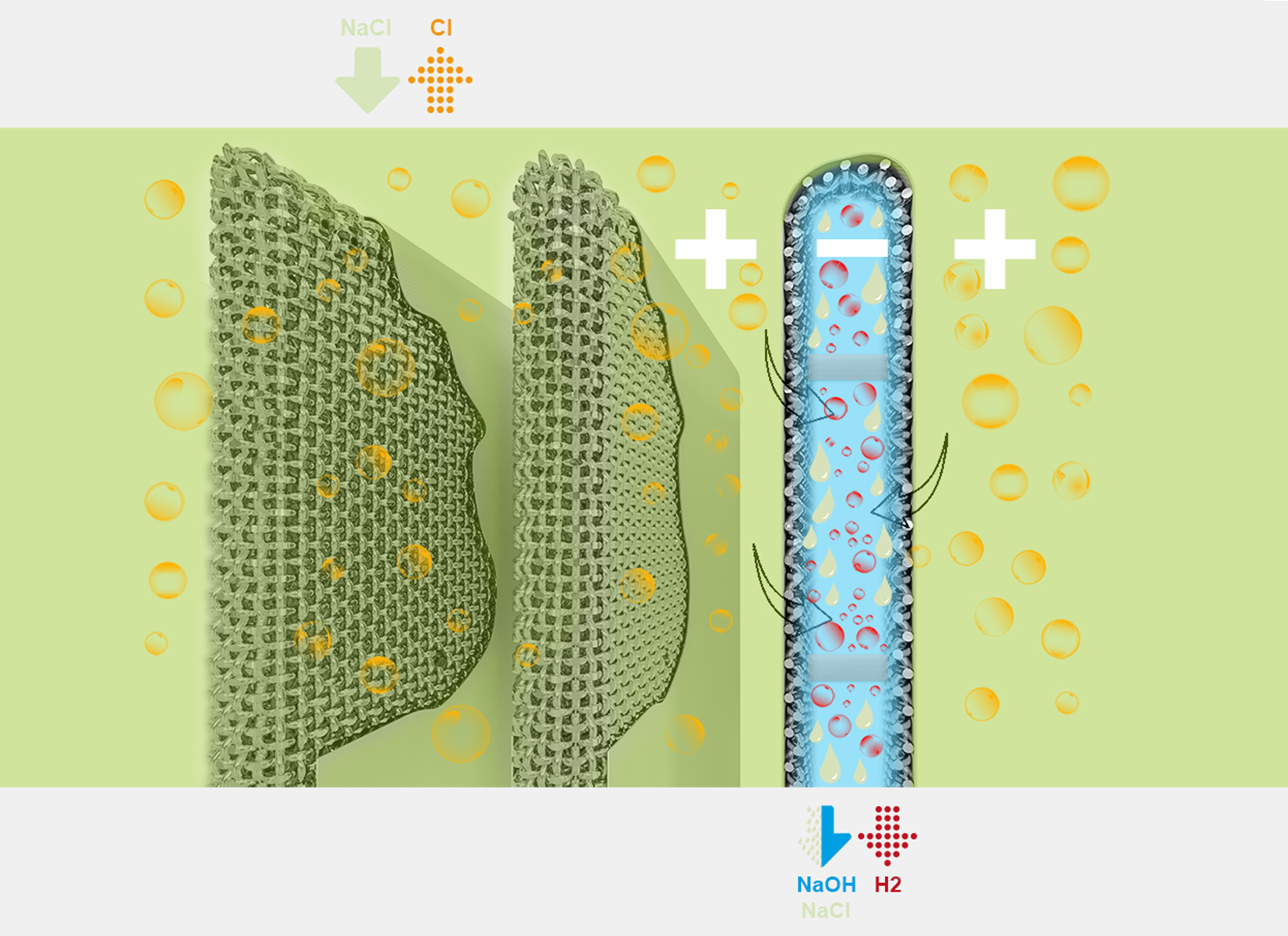
TELE METALLICHE PER CELLE A DIAFRAMMA PER PRODUZIONE DI SODA CAUSTICA
Le tele metalliche di Costacurta vengono utilizzate nel processo cloro-soda all’interno delle celle a diaframma.
In questo processo, per impedire che il cloro che si sviluppa all’anodo venga a contatto con il catolita, il comparto anodico è tenuto separato da quello catodico da un diaframma permeabile agli ioni ma impermeabile alle molecole di Cl2.
La soluzione di cloruro di sodio (anolita) viene inserita nel comparto anodico e fluisce, sotto battente idraulico, in quello catodico.
Nel primo comparto, quindi, gli ioni Cl– vengono ossidati e si sviluppa cloro gassoso. Nel secondo si formano invece idrogeno ed idrossido di sodio (NaOH soda caustica).
I gruppi catodici delle celle a diaframma (griglie di fondo e le cosiddette ‘dita catodiche’) sono realizzati con una tela metallica speciale in acciaio al carbonio prodotta da Costacurta.
Per la produzione di soda caustica, Costacurta è in grado di fornire pannelli piani a misura o i singoli elementi sagomati e saldati (dita catodiche) secondo le indicazioni tecniche e i disegni costruttivi del cliente.
Caratteristiche distintive delle tele metalliche Costacurta per queste tipologie di macchine sono:
- Fili di trama opportunamente laminati così da conferire alla tela metallica una superficie relativamente piana ed uniforme
- Spessore costante e controllato della tela metallica
- Elevata qualità del materiale impiegato e del processo produttivo







